Semiconductor & Consumer Electronics






We practice the semiconductor industry "Copy Exact" (CE) methodology. We are experts in tackling the primary enemies such as particulate contamination, outgassing, and thermal expansion.


Dimensional Tolerances & Surface Finish
Sub-Micron Machining: Tolerances must consistently hold between ±0.0001” to ±0.0005” (±0.0025 mm to ±0.0127 mm).
Extreme Flatness: Wafer chucks and optical components require flatness measured in light bands (e.g., $\lambda/4$) or down to 0.001 mm.
Mirror Finishes: Surface roughness (Ra) must hit 0.05 - 0.2 μm for vacuum sealing surfaces and photolithography components to eliminate micro-cavities where particles could hide.
Specific Material Capabilities
Metals: Expertise in machining brittle and exotic materials. Requirements include 316L Stainless Steel (Vacuum Induction Melted/Vacuum Arc Remelted [VIM/VAR] for ultra-high purity), Titanium alloys, Aluminum 6061-T6, and Hastelloy (C22/C276) for corrosive gas delivery systems.
Plastics: Complete mastery of ultra-clean, high-temperature polymers. Must machine and mold Polyimide (PI, continuous use >300°C), PEEK, PPS, and PTFE. These materials must be certified for trace-metal limits (often <1 ppm for elements like Copper, Zinc, and Iron) to prevent ionic contamination of wafers.
Environmental & Quality Baselines
Manufacturing, assembly, and packaging must occur in ISO Class 4 to 7 Cleanrooms with HEPA filtration (99.99% efficiency for 0.1 μm particles).
Compliance with SEMI standards (e.g., SEMI S2/S8) and rigorous Airborne Molecular Contamination (AMC) control protocols.
Post Processing
Type III Hard Anodizing (plasma resistance), Electropolishing (UHP stainless components), and ultrasonic precision cleaning.
Automotive






We thrive in the automotive manufacturing with highly regulated risk management and our ability to maintain absolute precision across massive production volumes.
Environmental & Quality Baselines
IATF 16949:2016 certification. This is the operational backbone for defect prevention and waste reduction.
Compliance with VDA 6.3 process audits (always audit-ready as German OEMs).
Process Capability & Traceability
PPAP Level 3 (Production Part Approval Process): The facility must be capable of generating comprehensive default submissions, including Design Records, Control Plans, and Material Test Reports.
Statistical Process Control (SPC): Unlike other industries where checking the final part is enough, automotive requires statistical proof that the process itself is flawless. Machining and molding processes must consistently demonstrate a Cpk > 1.33 (standard) or Ppk ≥ 1.67 for Special/Critical Characteristics (SC/CC).
Traceability: Full digital tracking from raw material heat numbers to final shipment to maintain a target defect rate of 0 PPM (Parts Per Million).
Specific Material Capabilities
Metals: High-speed, high-volume CNC turning and milling of high-strength dual-phase steels, lightweight Aluminum alloys, and Zinc/Magnesium for powertrain and chassis components.
Plastics: Molding of Glass-filled Polypropylene (PP), ABS, and Polycarbonate. For EV (Electric Vehicle) battery systems and under-hood components, thermal management requires expertise in molding high-temp polymers like PEEK and PPS that withstand environments exceeding 120°C and aggressive automotive fluids.
Medical Grade Capabilities


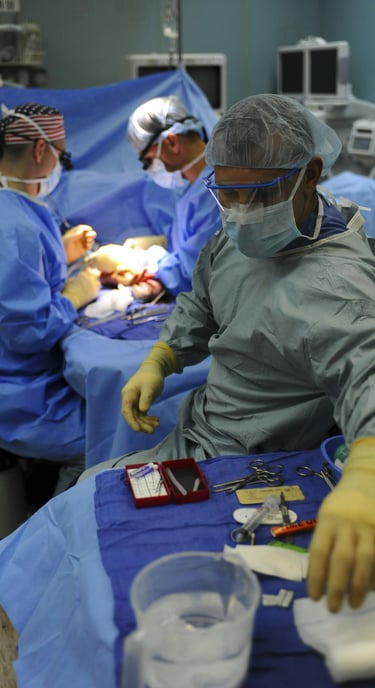
We govern our medical manufacturing by strict biological safety, traceability, and the physical stresses of repeated sterilization.
Environmental & Quality Baselines
ISO 13485:2016 certification and FDA 21 CFR Part 820 (cGMP) compliance.
Proactive risk management integration and complete UDI (Unique Device Identification) compliance for lifetime part traceability.
Biocompatibility & Sterilization
All raw materials must pass ISO 10993 and USP Class VI biological reactivity testing (assessing toxicity, intracutaneous reactivity, and implantation responses).
Materials and final assemblies must withstand aggressive sterilization methods without mechanical degradation or discoloration: Autoclave (steam up to 134°C), Ethylene Oxide (EtO), and Gamma Radiation.
Dimensional Tolerances & Surface Finish
5-Axis machining capabilities maintaining tolerances of ±0.005 mm.
Surface finishes below 0.2 Ra microns. Crucially, the machining process (feeds, speeds, and tool paths) engineered to prevent subsurface micro-cracks and residual stress, which harbor bacteria or cause catastrophic failure during repeated sterilization cycles.
Specific Material Capabilities
Metals: We machine medical-grade Titanium (Ti-6Al-4V ELI) for orthopedic implants due to its osseointegration properties (bone ingrowth). Also 316LVM Stainless Steel and Cobalt-Chrome alloys for surgical instrumentation.
Plastics: Liquid Silicone Rubber (LSR) molding, Implantable-grade PEEK (which mimics the modulus of human bone to prevent stress shielding), UHMWPE (for joint replacements), and specialized X-ray opaque (XRO) plastics for image-guided interventions.
Contact
Reach out for free consultation.
Phone
ADDRESS
Vietnam
+84 365531261
China
+86 186 2504 2992
Malaysia
+6013 7400321
© 2025. All rights reserved.
Vietnam
Lot CN2-4 and CN9-7, Yen Phong Expansion Industrial Park, Yen Phong Commune, Yen Phong District, Bac Ninh Province, Vietnam.
Malaysia
1-2-16, Tingkat Bukit Kecil 3, The Sun, 11900, Bayan Lepas, Pulau Pinang, Malaysia.
